When a ground state atom is collided with light of appropriate wavelength, the atom absorbs the light and enters an excited phase. The disadvantages of this method include a huge cost of infrastructure maintenance and operating expense, the presence of several emission lines or spectral interferences and the necessity of having samples dissolved in solutions. These include excellent linear dynamic range and limit of detection, low chemical interference, multi-element capability as well as a stable, reproducible signal. There are a few advantages of inductively coupled plasma atomic emission spectroscopy (ICP-AES). The results from such studies help in determining the life of the oil, as well as assist in quality control and help in functional efficiency of automobile engines.Īdvantages and disadvantages of ICP Atomic Emission Spectroscopy ICP-AES is also used for analyzing motor oils.The metal composition of two types of soil samples can be compared to determine the origin of the soil samples taken from crime scene. Forensic experts use this method to study soil samples found at the crime scenes and ascertain their origin. ICP-AES is frequently used for analyzing trace elements present in the soil.Inductively coupled plasma atomic emission spectroscopy is used to determine the presence of arsenic in food, metals in wine and to study trace elements that are bound to proteins.The various uses of ICP-AES have been discussed below: Uses of Inductively Coupled Plasma Atomic Emission Spectroscopy Inductively coupled plasma atomic emission spectroscopy (ICP-AES) employs the use of inductively coupled plasma for producing excited ions and atoms that radiate electromagnetically charged particles at wavelengths characteristic to a definite element. Inductively Coupled Plasma Atomic Emission Spectroscopy (ICP-AES) Light is emitted at a wavelength characteristic to each element which is then dispersed by a prism or grating and detected in spectrometer.įlame emission spectroscopy is frequently used while studying alkali metals for pharmaceutical research and analysis. 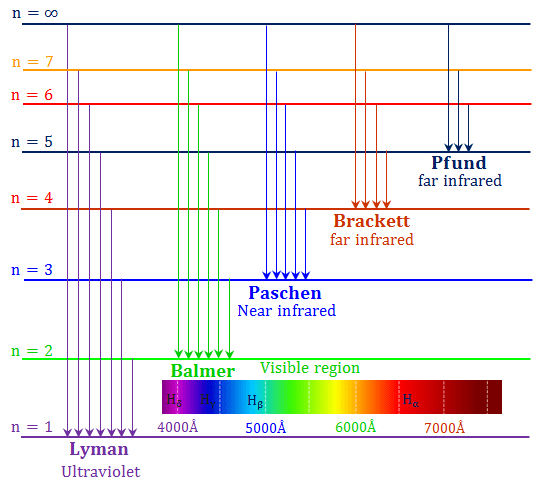
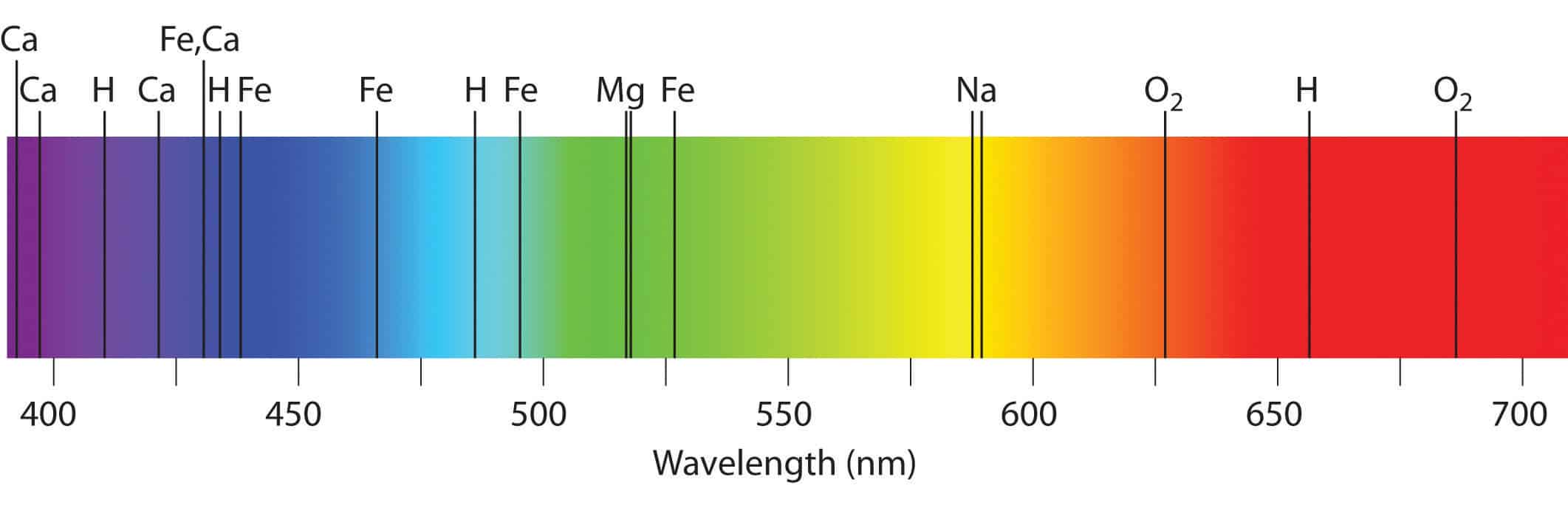
The heat also changes the atoms into electronically charged particles which emits light when they get back to the ground electronic state. Free atoms of the material are produced when the flame heat evaporates the solvent and breaks the chemical bonds of the analyte. In this method, a sample of the material to be analyzed is brought into flame in the form of a sprayed solution or gas. A study of these wavelengths can reveal the elemental structure of the sample.  
A characteristic set of wavelengths is emitted by each element or substance which depends on its electronic structure. The theory or working principle of Atomic Emission Spectroscopy involves the examination of the wavelengths of photons discharged by atoms and molecules as they transit from a high energy state to a low energy state. The atomic spectral line wavelength identifies the element and the intensity of light is proportional to the atom count of the element.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
